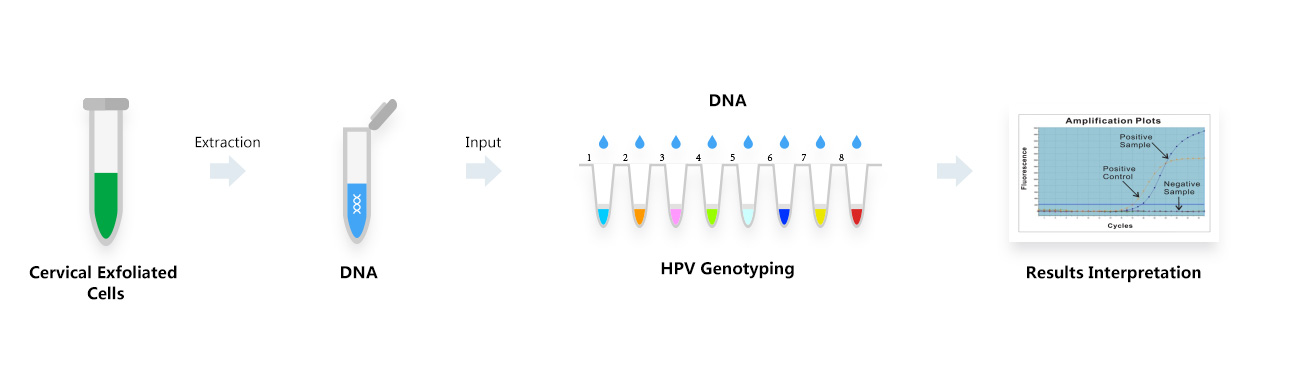
Cervical cancer is one of the common malignant tumors in women, and its prevalence is second only to breast cancer. There are about 130,000 new cases of cervical cancer and 30,000 deaths in China every year, which seriously threatens the health of our female population.
High-risk HPV testing and its combination with cervical cytology can significantly reduce the rate of missed diagnosis of cervical cancer and related lesions. More than 200 types of HPV have been identified, and persistent infection with high-risk HPV is the main cause of cervical cancer. Studies have shown that HPV infection can be detected in 99% of cervical cancer patients' cervical exfoliated cell samples, with HPV type 16 alone having a prevalence of more than 60% in this group. Low-risk HPV is highly associated with diseases such as flat warts and genital warts.
Several guideline consensus from WHO, FDA, ASCCP (American Society for Colposcopy and Cervical Pathology), IARC (International Agency for Research on Cancer) and other organizations recommend HPV testing for the female population aged 30-65. The 2017 edition of the Expert Consensus of the Expert Committee of the Colposcopy and Cervical Pathology Branch of the Chinese Eugenics Science Association clarifies the process of handling clinical cervical cytology, HPV testing and the combination of the two in China.
HPV testing can indicate the risk of recurrence after treatment in people with cervical cancer and related lesions. The chance of HPV infection in a person's lifetime is as high as 80%, and such people are at risk of re-infection or persistence of pre-infection after treatment, while high-risk HPV-infected people are at high risk of recurrence, so it is very important to monitor the dynamics of the HPV virus in the post-treatment population.
HPV testing can guide cervical cancer vaccination. In recent years, a variety of cervical cancer vaccines produced by different manufacturers have come on the market, and each vaccine does not target exactly the same HPV type. In order to achieve better immunization effect, you should test your own HPV infection status before vaccination, and choose reasonably for your own situation.





 0752-2228195
0752-2228195